Article, news, WORLD
Decades in the Making, First Malaria Vaccine Approved by WHO
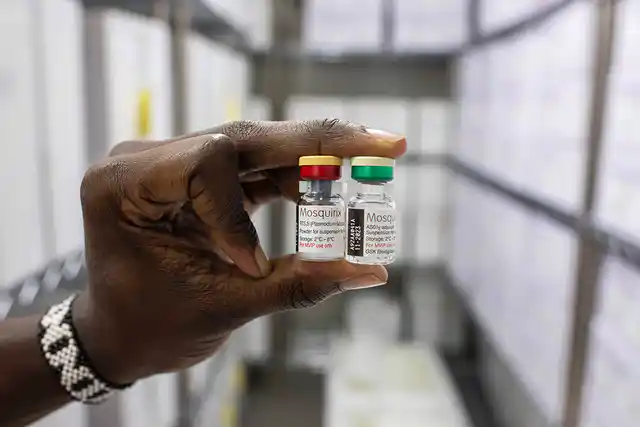
The World Health Organization recently made a groundbreaking development in global health as they approved the first vaccine for malaria in Africa. Malaria is responsible for over 400,000 recorded deaths a year, with a significant amount of cases occurring in children under the age of five. Additionally, the disease has historically contributed as a significant economic inhibitor to less economically developed areas of the world, particularly in Africa.
Africa’s state in the modern world has long been affected by the shadow of the disease, as the continent shoulders half of the world’s cases annually. The development of an effective vaccine symbolizes the potential for more Africans to achieve a higher quality of life going into the future.
Malaria was first identified and named over 130 years ago and has stalked humanity as one of its major diseases for millennia. Despite this, an effective remedy has long eluded medical professionals. The vaccine makes its target the most deadly and common strain of the virus, Plasmodium falciparum.
“We have long hoped for an effective malaria vaccine, and now for the first time ever, we have such a vaccine recommended for widespread use. Today’s recommendation offers a glimmer of hope for the continent which shoulders the heaviest burden of the disease, and we expect many more African children to be protected from malaria and grow into healthy adults,” said Dr. Matshidiso Moeti, the WHO Regional Director for Africa.
Malaria Vaccine Has Been Decades In The Making
The Vaccine, RTS, S/AS01 with brand name Mosquirex, was developed by PATH Malaria Vaccine Initiative and British pharmaceutical giant GlaxoSmithKline (with additional funding by the Bill and Melinda Gates Foundation) and required four injections in order to be effective. The vaccine has been gestating in a clinical trial period since 2015 and was approved by the WHO just recently. The trial period involved injecting over 800,000 people with the vaccine.
The seeds of the effort, however, reach over a much larger span of time, reaching back to 1987 when GlaxoSmithKline first began attempts to develop a remedy. Producing an effective agent has subsequently taken millions of dollars in research and development.
The length of time that it has taken the medical community to develop an effective response to Malaria might seem surprising given the severity of the disease and its impact on society. This time frame also seems conspicuous in light of the recent development of the COVID-19 vaccine, which was produced at unprecedented speed in about a year. Doctors stress this is due to the complex nature of the malaria virus.
Issues Remain With Outreach, Efficacy
Although the vaccine represents a beacon of hope for Africa, some concerns have been raised in the medical community over deploying a vaccine that has a somewhat underwhelming efficacy rate. In severe cases of Malaria in young children, the vaccine was shown to have an efficacy of about 30% after all four doses. Although this is relatively low, it still contains the potential to save tens of thousands of lives, as there was a 70% reduction in hospitalization or death in those who had received the medication.
Medical professionals in Africa stress that public outreach and communication between local governments and their populaces will be essential in order for the true potential of the vaccine to be utilized, especially since efforts will be made underway under the shadow cast by the misinformation scandals that have surrounded the COVID-19 vaccine.
“The misinformation around COVID-19 vaccines should teach us that we can’t take community trust for granted,” states James Tibenderana, a Ugandan epidemiologist who works at the Malaria Consortium in London.
Additionally, disappointment has been expressed over a perceived double standard over a Vaccine that is primarily intended to be used by people in the Global South, with some pointing out that the efficacy rates of the drug would not necessarily be acceptable in wealthier western nations.
“I don’t think a 30% effective vaccine would be acceptable for Americans,” says Badara Cisse, a researcher at the Institute for Health Research, Epidemiological Surveillance, and Training.
Developing A Path Forward
Although the vaccine results are modest, it contributes to building a foundation towards fighting the disease in the future. Some, such as WHO Director Dr. Tedros Adhanom Ghebreyesus, have touted the event as “A historic moment.”
Professionals insist the vaccine will not replace existing prevention and remediation methods but instead work in tandem with those efforts in the ongoing struggle to contain the virus. GlaxoSmithKline has committed to widespread distribution and will supply up to 15 million doses a year at no more than 5% above the cost of production.
Despite reservations about the vaccine’s overall efficacy, it does show favorability in several key areas, including carrying no negative interactions with other routine vaccines. Having an at least somewhat viable vaccine also primes the pump for the future development of even more effective remedies.
Gareth Jenkins, director of advocacy at the charity Malaria No More, pointed to the obligation of wealthier nations to continue investing in the research that produced the vaccine and added that if one could ‘’finally bring about a zero-malaria world’’ it would benefit wealthier nations as well.
“If we save lives from malaria today, we can also protect ourselves against the diseases of tomorrow.

